News Hub
The latest breaking stories
Access up-to-date news and information on a wide range of press releases including business, finance, technology, healthcare, entertainment, and more.
Discover the most important & comprehensive stories delivered directly to you daily.

- Energy, Environment
- 04/03/2026
- 15:10
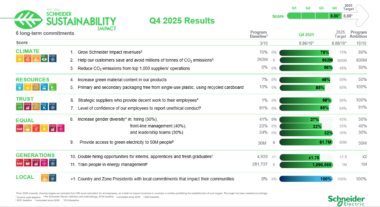
Schneider Electric completes its Sustainability Impact (SSI) 2021-2025 program, setting the stage for the next chapter
Sydney, Australia March 4, 2026– Schneider Electric, a global energy technology leader, has published its 2025 extra-financial results, marking the conclusion of its Schneider…
- Contains:


- Medical Health Aged Care
- 04/03/2026
- 14:20
Meet Charlie, Westmead Private Hospital’s newest team member
Key Facts: 10-year-old golden retriever Charlie joins Westmead Private Hospital as a therapy dog, accompanied by owner Julie Quinn The therapy dog program aims…
- Contains:
- Information Technology
- 04/03/2026
- 13:58
Infosys and Intel Deepen Strategic Collaboration to Unlock AI Value for Enterprises Globally
Infosys and Intel Deepen Strategic Collaboration to Unlock AI Value for Enterprises Globally Focus on scaling production‑ready deployments, optimizing performance, and delivering measurable enterprise outcomes across industries with Infosys Topaz and Intel’s high-performance compute platforms AUSTRALIA – March 04, 2026: Infosys (NSE, BSE, NYSE: INFY), a global leader in next-generation digital services and consulting, and Intel, a global leader in computing innovation, today announced the next phase of their strategic collaboration to help enterprises move from AI pilots to production at scale. The expanded collaboration brings together Intel’s high‑performance, energy‑efficient compute platforms and Infosys Topaz Fabric – a purpose-built agentic…


- Building Construction, Women
- 04/03/2026
- 13:56
Women show the way in steel: ASI recognises International Women’s Day on March 8
Shaking off gender bias hasn’t been easy in the steel industry. Until recently, the prevailing view was “husky men doing dirty work”. That has…
- Contains:


- Fashion, Women
- 04/03/2026
- 13:28
Pillow Talk unveils a new era of comfort with its Autumn 2026 collection
An elevated edit of quilt cover sets, plush cushions and accent furniture in warm, earthy tones heroes this season’s campaign, designed to ground spaces…
- Contains:
- CharitiesAidWelfare, International News
- 04/03/2026
- 13:22
Thousands of Lebanese children carrying fear instead of schoolbags as renewed hostilities displace 55,000
Key Facts: Over 12,000 families (55,000+ people) have been displaced in Lebanon due to renewed hostilities, with one million children needing humanitarian aid World Vision has provided emergency supplies and hot meals to 5,552 displaced people, adapting school snack programmes for children in shelters Children face increased risks including psychological distress, disrupted education, family separation and poor living conditions in overcrowded shelters World Vision is calling for immediate de-escalation and protection of civilians, particularly children who need psychosocial support and education The organisation has been working in the Middle East for over 50 years, providing emergency assistance and long-term development…
- Contains:
- Information Technology
- 04/03/2026
- 13:10
Open Campus, Le & Associates, and SKALE partner to pilot credential-powered job matching initiative in Vietnam
HO CHI MINH CITY, Vietnam, March 04, 2026 (GLOBE NEWSWIRE) -- Open Campus, the community-led decentralized autonomous organization (DAO) building the blockchain-powered financial layer for education, today announced that it has entered into a Memorandum of Understanding (MoU) with Le & Associates and SKALE in relation to the intended launch of a credential-powered job matching initiative focused on the Vietnamese market.The collaboration will combine Open Campus’s education ecosystem and credential and identity infrastructure with Le & Associates’s recruitment expertise and SKALE’s digital recruitment platform to enable candidates to share verified achievements directly with employers, and allow companies to screen applicants…


- CharitiesAidWelfare, Entertainment
- 04/03/2026
- 12:16
Canberra it’s time to show us your personali-tea!
Key Facts: Cancer Council ACT’s Australia’s Biggest Morning Tea 2026 will be held throughout May and June, with the official date on Thursday 21…
- Contains:


- Business Company News
- 04/03/2026
- 11:34
Local businesses rally for mental health: Annual golf day set to support Lifeline Gippsland.
Final spots available as Latrobe community prepares for charity fundraiser at Yallourn Golf Club LATROBE CITY, VIC – The Latrobe City Business Chamber will…
- Contains:
Media Outreach made fast, easy, simple.
Feature your press release on Medianet's News Hub every time you distribute with Medianet. Pay per release or save with a subscription.

